Walking around Europe’s largest cannabis manufacturing facility, it quickly becomes obvious that I’m in the hands of a veteran. Even though we’re surrounded by millions of euros' worth of state-of-the-art machinery, Somai Pharmaceuticals’ founder and CEO Michael Sassano is more concerned with the tiny scuffs he’s noticing on the walls and floor than the impressive technology.
“It’s a little rough,” he says, dropping down to his knees to examine the easy-clean epoxy surface. Sassano has spotted a tiny dimple in some of the sealant connecting two panels. It’s almost indiscernible to the untrained eye but, now, building his 16th cannabis manufacturing facility, he’s hyper-aware of anything that might slow the operation down later along the line.
“It’s the details that’ll sink you, brother,” he says, in a drawling American accent.

“A bit of roughness like that: run a machine over it a few hundred times, that’s going to be a full-on scratch — a breeding ground for bacteria. Once that happens, you’ll have to shut the whole operation down while you fix it.”
This high level of attention to detail is understandable, given the stakes. Hundreds of startups are seeking to get their noses out in front in the European medical cannabis market which is expected to grow from $4.96bn in 2022 to $13.4bn in 2027.
Sassano says that the majority of these startups are in cannabis cultivation, and they'll become natural clients for Somai. His company turns the raw cannabis plant into medical products like sprays, drops, vape liquids, softgels and transdermal patches (“a doctor would rather not prescribe you a joint to smoke,” he explains).

On the production line
Even though the manufacturing line, located 30 minutes outside of Lisbon, isn’t fully operational on the day of my visit, we’re still required to suit up in antibacterial smocks, shoes and hairnets. When you’re making medical products to the highest certification, keeping contamination out of the production is priority number one.
“We’re glorified cleaners,” Sassano jokes. But his dry humour doesn’t cover for the impressiveness of the technical operation. Built within a pretty standard-looking warehouse from the outside, Somai’s production line is a U-shaped procession of airlocked and pressurised chambers, each housing its own piece of specialised equipment.
After the raw “biomass” (the cannabis buds) enters the warehouse, it’s first cleaned. Somai’s managing director Anton Nakhodkin is visibly stressed describing this unsanitary part of the process.
“It is extremely dusty,” he frets.

After the biomass is cleaned and weighed, we’re onto the serious machinery.
To be turned into medical products, the cannabis buds must first be turned into an oil.
This is where the cryo-ethanol extractor comes in. The machine takes in cleaned biomass and runs it through a process using ethanol chilled to minus 40C, to extract crude oil from the plant material.


Once this cannabis oil has been obtained, it has to be purified. It’s wheeled next door to meet the distillation machine (Sassano’s favourite toy in the facility). Here, dark crude oil at a purity of roughly 55% THC (the primary psychoactive ingredient in cannabis) is turned into a clear distillate with a THC purity which can range from 80-95%.
Next, the distillate is taken to meet the formulation room — the place where it’s turned into one of the 42 different products, including sprays and oils, that Somai intends to roll out.
Different products
“42 is extremely aggressive,” says Sassano. “That’s about 32 more than the nearest guy.”
Much of this product differentiation comes in the different mixes and concentrations of THC and CBD (another psychoactive compound in cannabis which is believed to reduce anxiety among other benefits) that Somai produces.
This balance changes the intensity and general effect of the medical product, while another factor, homogenisation, influences how quickly edible products are absorbed into the body’s system.
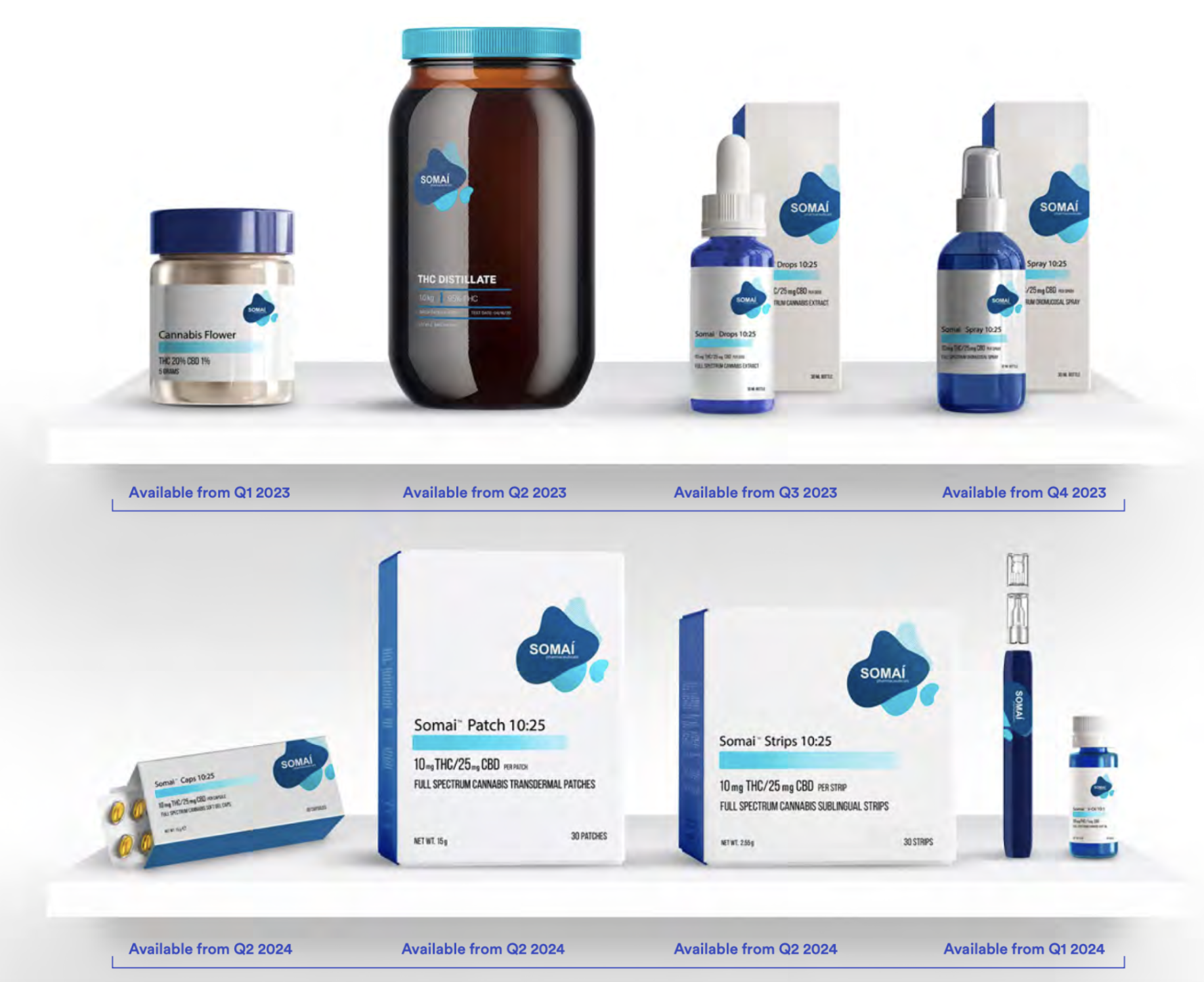
Sassano says that these differences can all make meaningful differences to the way a patient responds to medicinal cannabis, adding that taste will be another differentiation in Somai’s product line.
“There is no reason that your medicines should taste like crap,” he says.
After being formulated, the cannabis derivatives are then taken off to be bottled and packaged. Before sending out for distributors, all of Somai’s products are quality controlled on-site to ensure they match up to the highest GMP medical certification for cannabis. The certification ensures that medical products are consistent, high-quality and don’t degrade more quickly than stated.

The need
These 42 products will give doctors a wide range of options for treating different conditions in different people. In Europe, Sassano says, the average age of patients using medical cannabis is around the 50-55 mark — clearly showing that the drug isn’t only valued by hippies at music festivals, as the stigma suggests.
“The number one category of indication [currently being treated with cannabis] is pain,” Sassano explains. “Then you’ve got conditions like epilepsy, sclerosis and arthritis.”
While doctors can prescribe these cannabis products as a medicine today, the treatments are not yet approved through the clinical trial process. What this means is that a doctor can only say “this might help your arthritis” rather than “we are clinically certain this will help your arthritis”.
As well as producing medical products that can be prescribed, the company is also launching clinical trials to develop approved cannabis-based drugs for chronic pain and arthritis.
Sassano says that there are currently 200k insured patients in Germany using cannabis treatments, while less developed markets like the UK have around 17k patients, with that number “going rapidly up” every year.
The startup market
Sassano says that demand for cannabis in Europe is about to explode, with Germany looking set to legalise the drug completely in the coming years — a move that he says will supercharge European rollbacks of regulations.
“You have the real kindling of a brush fire going on right now,” he says. “If Germany goes, I imagine Portugal will be almost instantaneous.”
This, he says, is being accompanied by a general swathe of relaxation around medical cannabis regulations in countries like France, Spain and Switzerland.
Startups aren’t naive to what could be a massive increase in the size of Europe’s cannabis market in the coming years. Sassano says that there are currently hundreds of founders around Europe applying for growing licences to meet this demand.
“They all want to work with us,” he says. “We give them that end-to-end solution, and allow them to diversify their product so they don’t just have to sell the [cannabis] flower. What we make is a higher margin product.”

Manufacturing startups like Somai are in competition with big hitters from Canada, where the medical cannabis market is much more mature. But Sassano says that due to market woes back home, Canadian companies’ “capital is distracted”, with none of them having anywhere near as much manufacturing capacity in Europe as Somai.
Why Portugal?
Before launching the company in 2020, Sassano was CEO of US cannabis company Solaris Farms. He’s clearly relishing the chance to bring that expertise to Europe, where the market is younger and the competition less fierce.
“We come from the US market where we're in a much more competitive place. We’re used to much lower price targets, so we can easily achieve a much more profitable experience here,” he says.
As the biggest manufacturing facility on the continent, and a first mover, Sassano says that Somai will be generating €7m in sales in 2023, with that figure doubling in 2024 and climbing to €35m in 2025. He says that profitability should be achievable for the company by the end of next year.

The company’s already raised €22m in funding from a mix of private equity funds, family offices and angel investors, and Sassano says that he’ll be looking for banking partnerships to fund things like clinical trials and product registrations in different markets.
Portugal, the Somai founder says, is a perfect place to launch a cannabis business. That’s partly due to regulators who genuinely want the industry to succeed and partly due to a large pool of highly skilled and relatively inexpensive talent who work hard.
“I’ve got a brilliant team,” he says. “They work hard, they really care. And you know what? I really care as well.”